Join the cohort of leading companies

.png)
.png)
Calculate your impact in minutes
.png)
Clear, guided workflows
Use pre-built templates to guide you
User-friendly
Accessible to non‑experts as well as specialists
Built for pharma
Use several models and compare materials to identify the lowest-impact packaging designs
Download valuable reports fast
Generate clear, downloadable reports that work for internal teams, regulators and external stakeholders
Maximise impact. Minimise costs.
Reduced raw material procurement costs
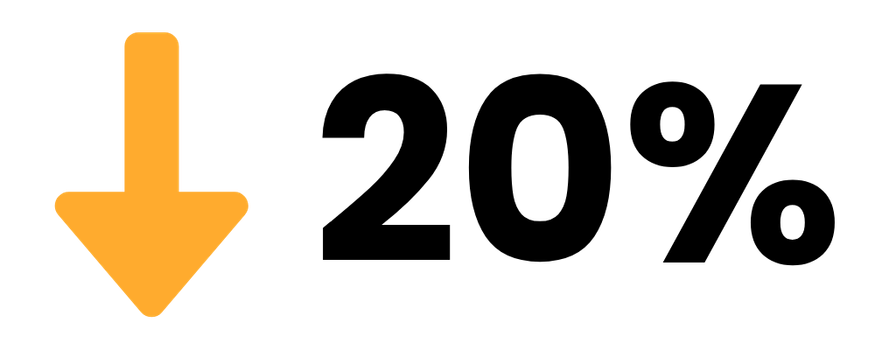
Shipping & Logistics
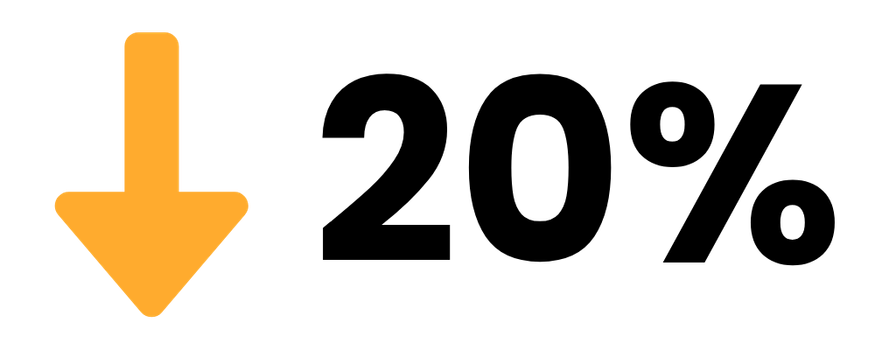
0 consultancy Fees
.png)
Packaging at a glance
What is happening in the industry
Voluntary Sustainability & Data
Complexity & Data-Driven Change
Long-Term Adoption Cycles
External communication
Generate corporate & product carbon footprints, LCAs, EPDs. Get data quality verification and AI-assisted ecodesign suggestions
.png)
Use the data generated in Pilario to comply with the increasing number of regulations and frameworks
.png)
Ensure your statements are backed by verifiable data
Generate EPDs and download customisable reports to respond to customer and investor data requests
.png)
Strategic supply chain management for net zero goals
Use CSV/Excel templates, automated feeds or API to import, create and update products
Automated data gap assessment through quality checks
Integrate with your ERP systems
Import existing supplier spreadsheets directly into Pilario or send data collection forms to suppliers and track responses and data completeness
.png)
Identify envrionmental hotspots and risks within your supply chain
Get AI insights into your portfolio
Compare alternative materials, model scenarios, analyse trade-offs
Quantify carbon and broader environmental impacts of each option
Compare economic costs
.png)
Frequently asked questions
In pharma, every change is strictly data-driven. An LCA is required to confirm a real environmental benefit in carbon footprint, water usage, and waste before a material is swapped. Crucially, LCA helps ensure that a packaging change doesn't accidentally shorten a medicine’s shelf life; if the Active Pharmaceutical Ingredient (API) is wasted due to poor packaging, the overall environmental impact is far worse than the packaging itself.
Pilario enables EPR readiness by providing a digital dashboard that automates the tracking of packaging material types and weights, allowing you to calculate modulated fees and comply with regulatory reporting effortlessly. Beyond current compliance, the platform supports long-term scenario planning by allowing your team to model "what-if" ecodesign changes—such as shifting from PVC to mono-materials—to quantify future CO2e reductions and economic trade-offs. This automated approach ensures your pharmaceutical packaging strategy remains resilient against evolving standards while protecting your business operations from future environmental risk.
Yes. Once a product model is created, it can be duplicated and adapted across formats, pack sizes or geographies, enabling portfolio-level analysis.




.png)
